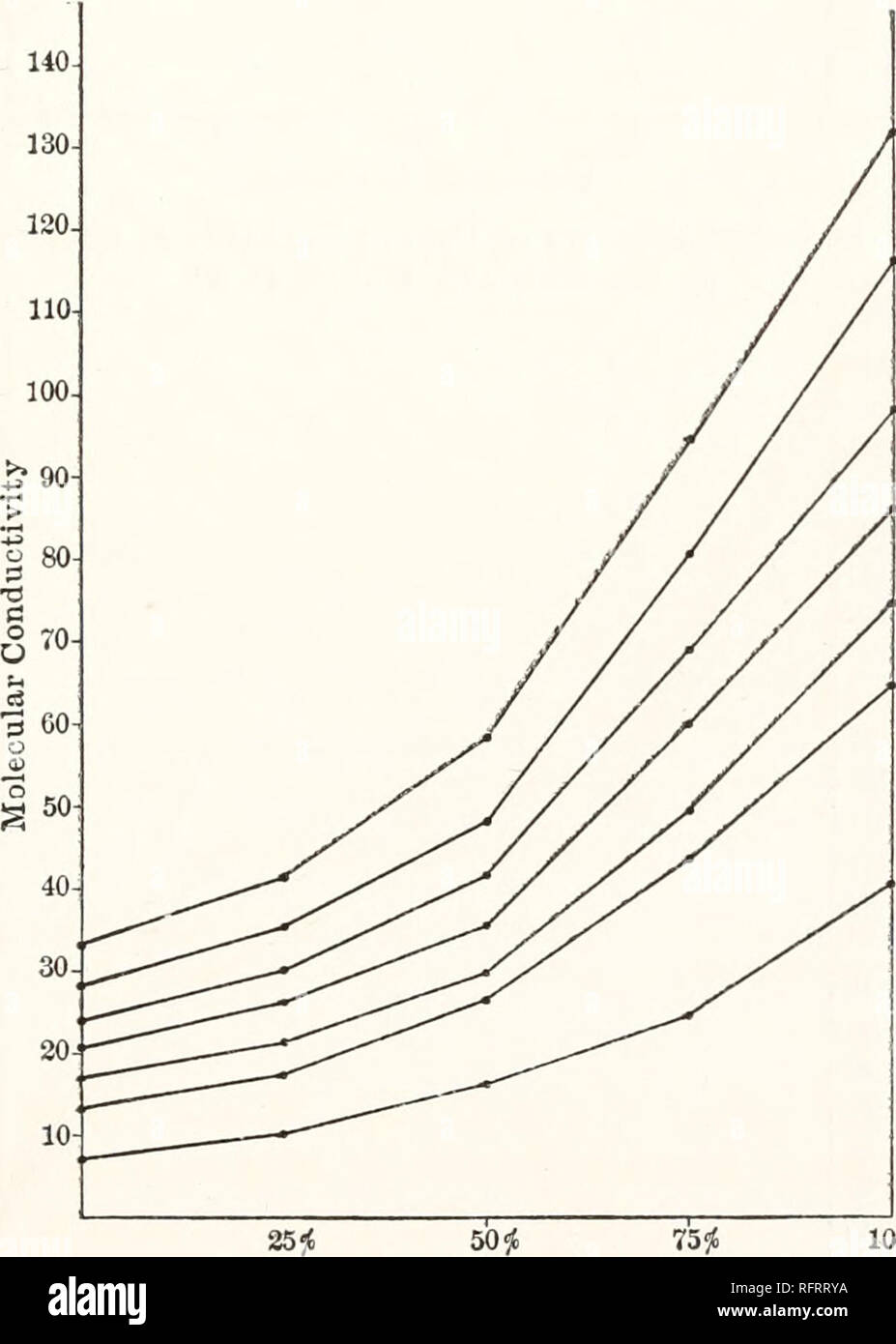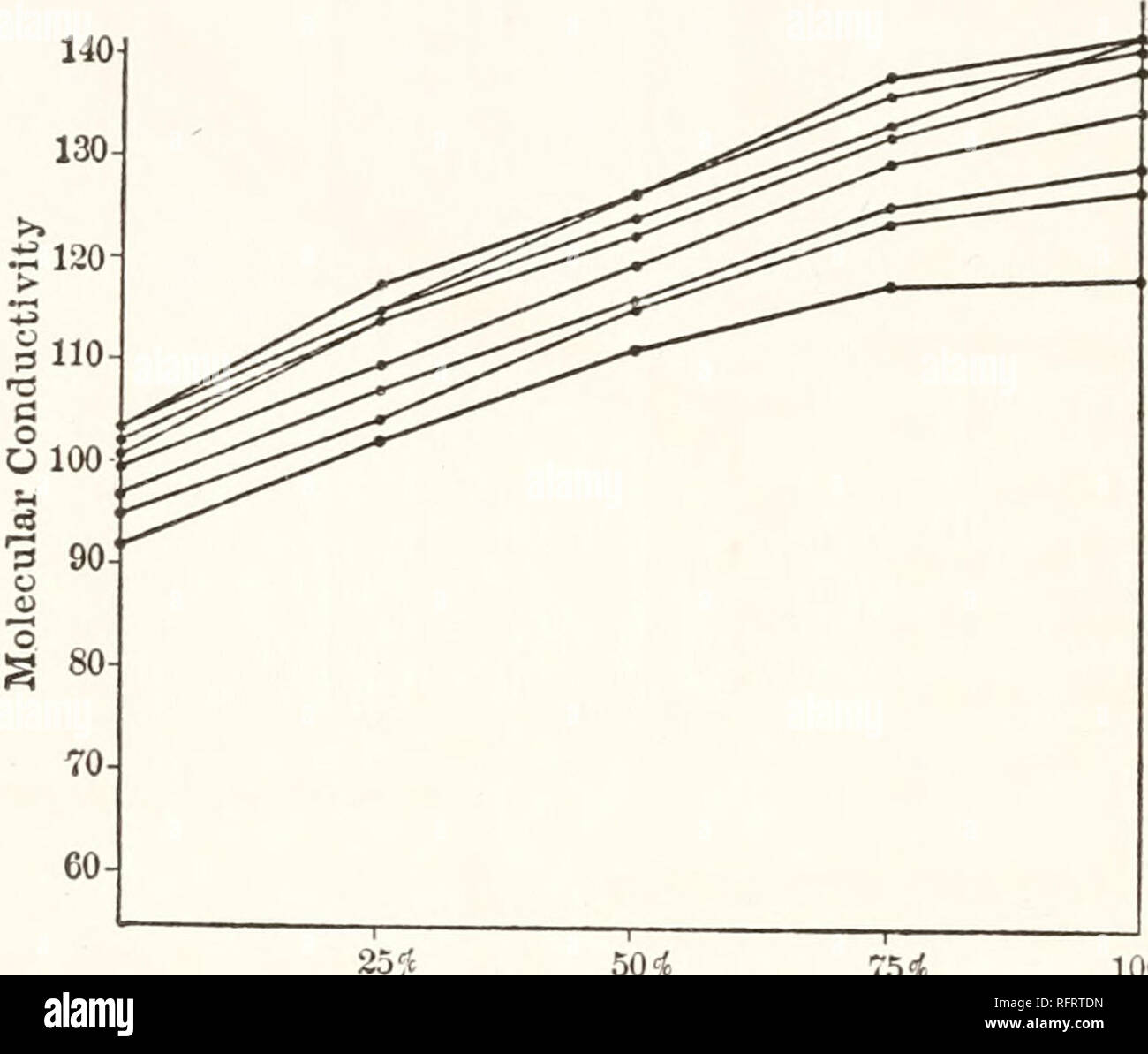
Carnegie Institution of Washington publication. 94 CONDUCTIVITY AND VISCOSITY IN MIXED SOLVENTS. Tables 55 and 56 (figs. 28 and 29), for potassium iodide in mixtures of acetone and methyl alcohol, show
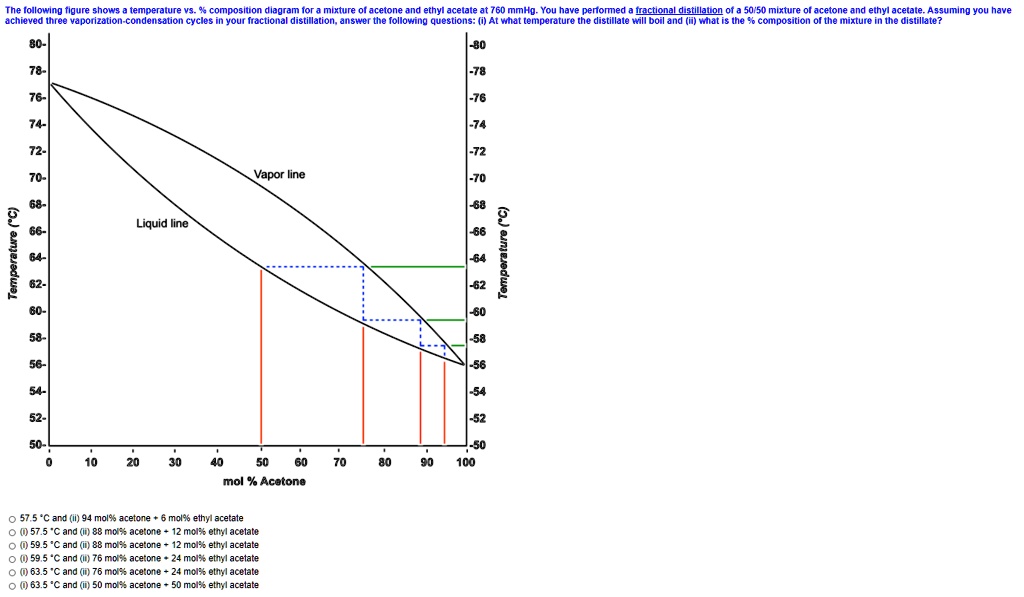
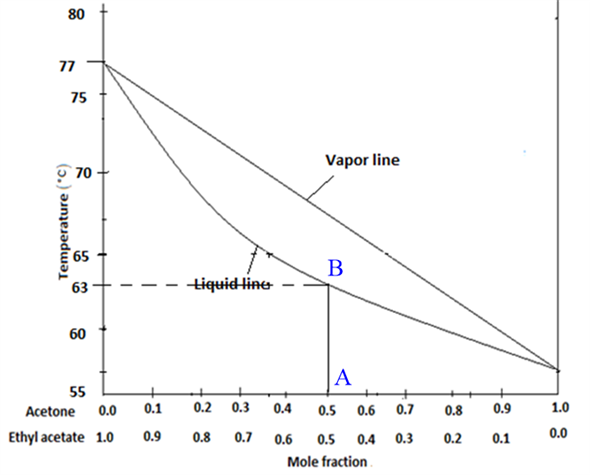
SOLVED:The lollowing tigure shows temperature comnosilon dladramor mixture Ol acetone and ethyl acetale 760 mmho You have penomed Ictlonal dltilalon of a 50/50 mixture acetone and ethyl acetale 4 9 qumino Vou
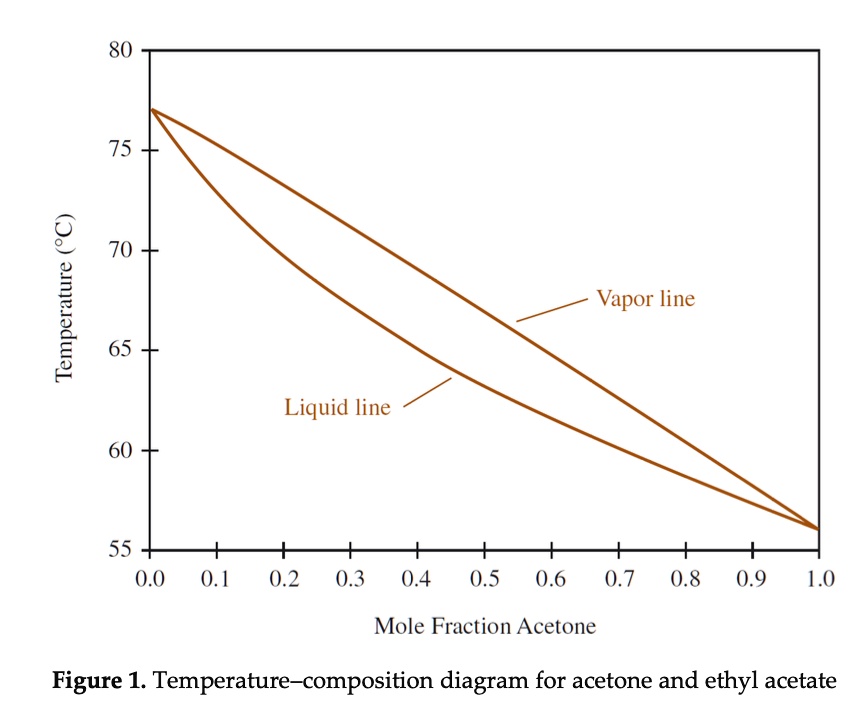
SOLVED:80 75 70 Temperature 65 Vapor line Liquid line 60 55 0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 Mole Fraction Acetone Figure 1. Temperature-composition diagram for acetone and ethyl acetate
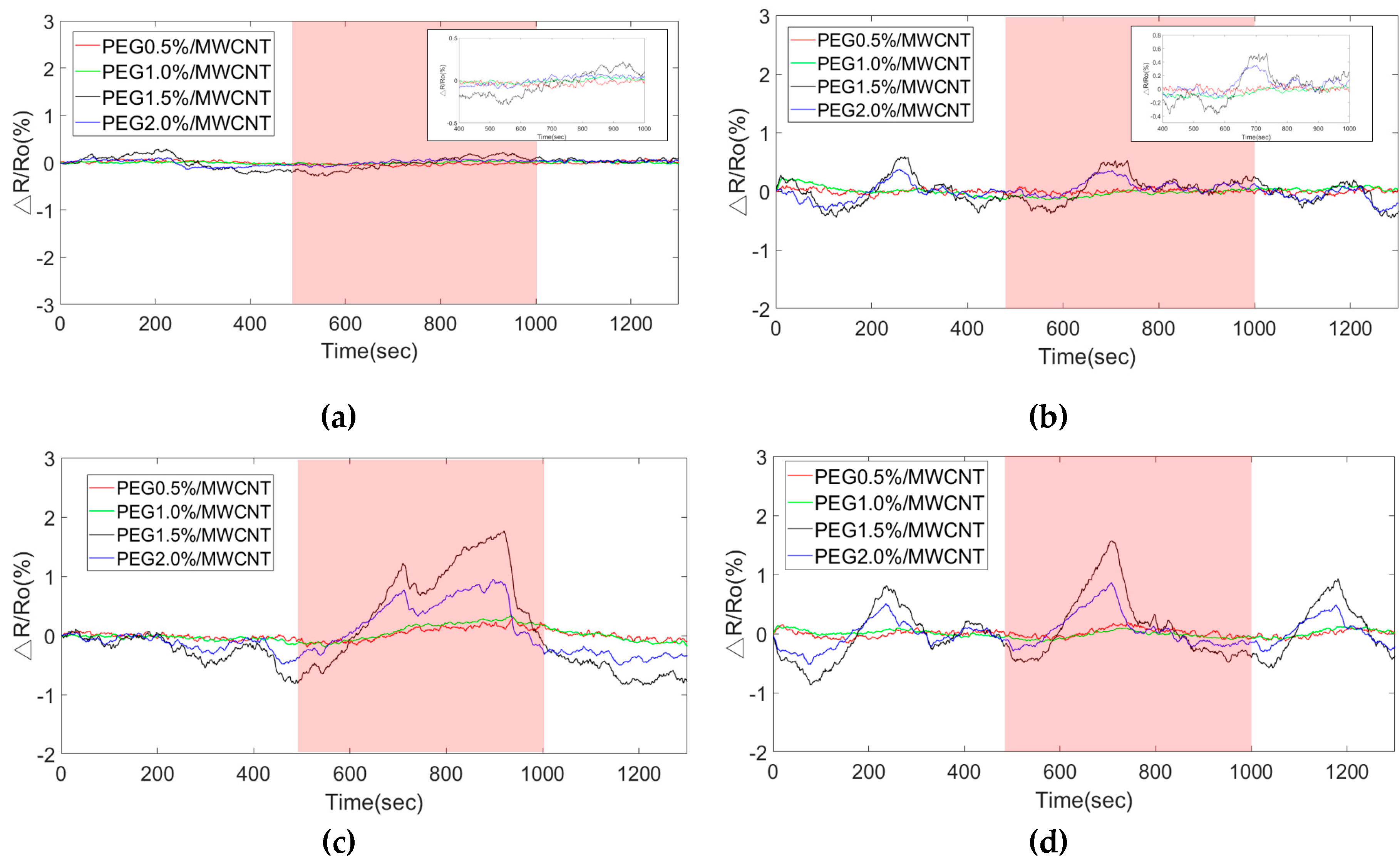
Polymers | Free Full-Text | Sensitivity Enhancement of Acetone Gas Sensor using Polyethylene Glycol/Multi-Walled Carbon Nanotubes Composite Sensing Film with Thermal Treatment | HTML
![PDF] The influence of pressure and temperature on the crystal structure of acetone | Semantic Scholar PDF] The influence of pressure and temperature on the crystal structure of acetone | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ea803c7523250d48aa3abd31ad12fe887bc8f63a/2-Figure2-1.png)
PDF] The influence of pressure and temperature on the crystal structure of acetone | Semantic Scholar

Interaction of Acetone with Single Wall Carbon Nanotubes at Cryogenic Temperatures: A Combined Temperature Programmed Desorption and Theoretical Study | Langmuir
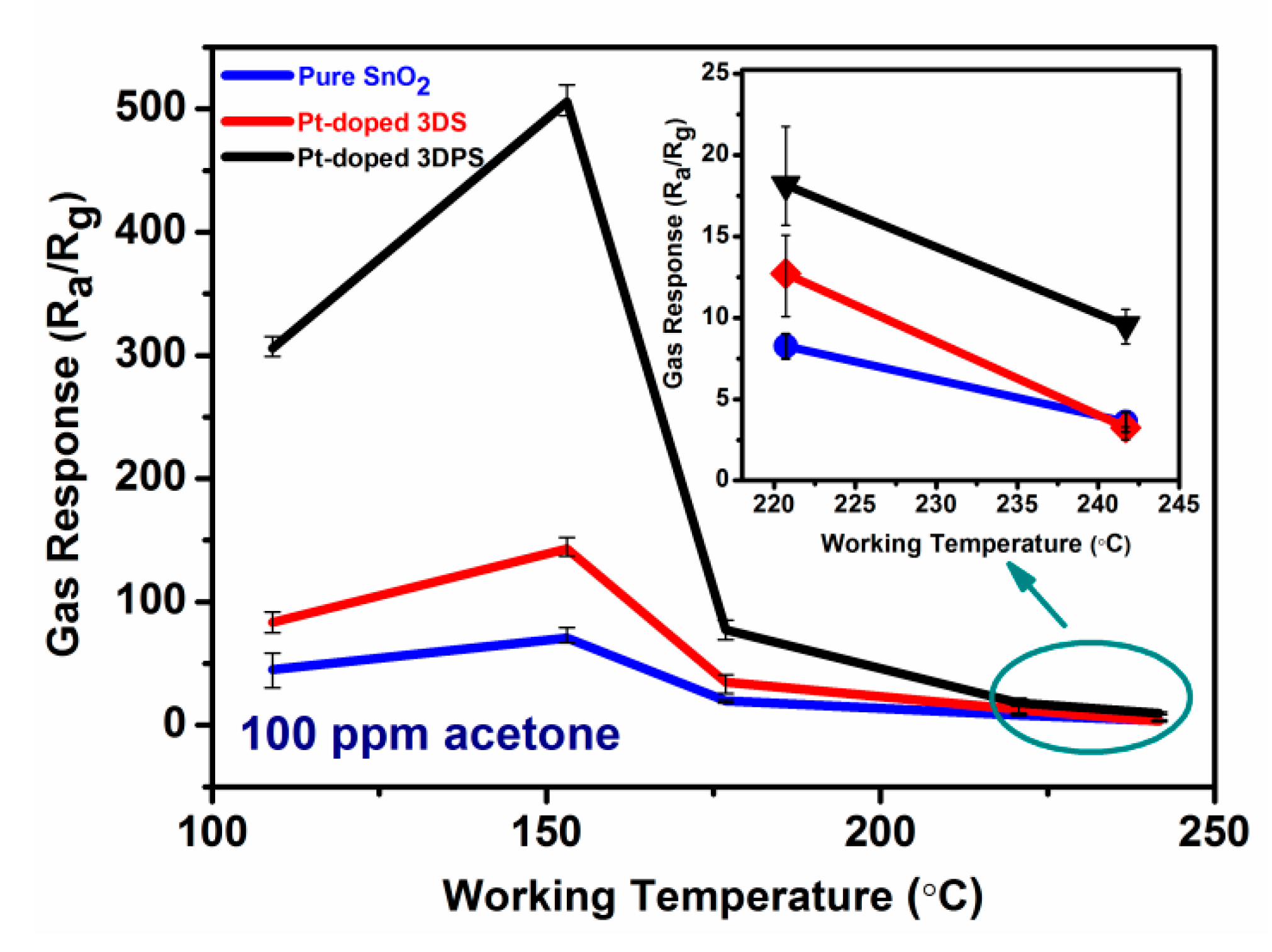
Sensors | Free Full-Text | A Highly Sensitive and Selective ppb-Level Acetone Sensor Based on a Pt-Doped 3D Porous SnO2 Hierarchical Structure | HTML
Equilibrium phase diagram of acetone/ethanol mixture under the pressure... | Download Scientific Diagram
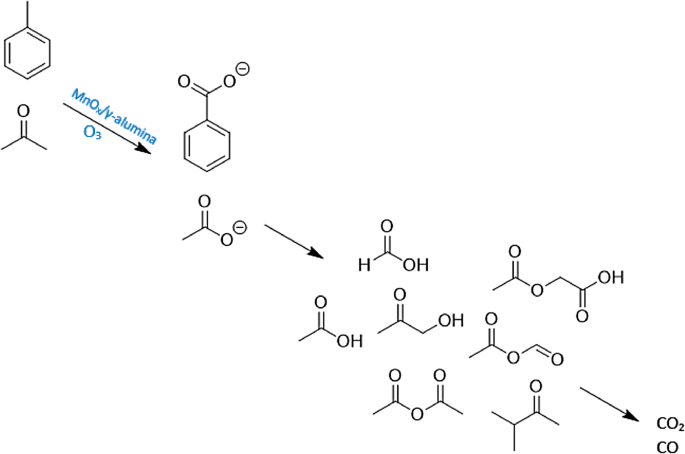
Low Temperature Catalytic Oxidation of Binary Mixture of Toluene and Acetone in the Presence of Ozone | SpringerLink
Measurements and modeling of acetone laser-induced fluorescence with implications for temperature-imaging diagnostics

thermodynamics - What mixing ratio of ethanol and acetone has the lowest freezing point? - Chemistry Stack Exchange
Sensing mechanism of ethanol and acetone at room temperature by SnO2 nano-columns synthesized by aerosol routes: theoretical calculations compared to experimental results - Journal of Materials Chemistry A (RSC Publishing)