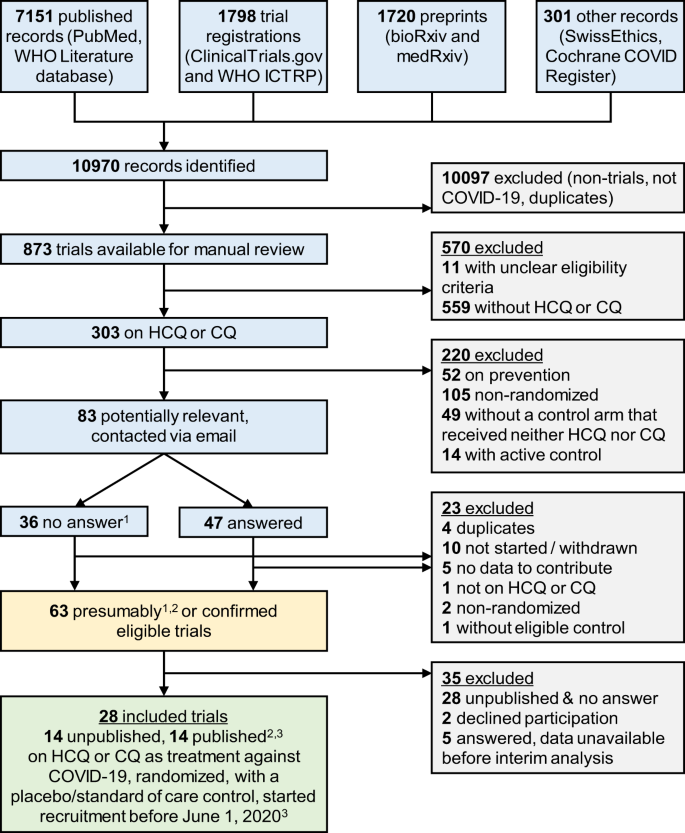
Missing clinical trial data: the evidence gap in primary data for potential COVID-19 drugs | Trials | Full Text

How to avoid common problems when using ClinicalTrials.gov in research: 10 issues to consider | The BMJ

The spectrum of activity, route of administration, production company,... | Download Scientific Diagram
Patient-reported outcomes and target effect sizes in pragmatic randomized trials in ClinicalTrials.gov: A cross-sectional analysis | PLOS Medicine

Standardisation of diet and exercise in clinical trials of NAFLD-NASH: Recommendations from the Liver Forum - Journal of Hepatology

Efficacy and safety of hydroxychloroquine as pre-and post-exposure prophylaxis and treatment of COVID-19: A systematic review and meta-analysis of blinded, placebo-controlled, randomized clinical trials. - The Lancet Regional Health – Americas

Publication of NIH funded trials registered in ClinicalTrials.gov: cross sectional analysis | The BMJ