References in Analyzing Spin in Abstracts of Orthopaedic Randomized Controlled Trials With Statistically Insignificant Primary Endpoints - Arthroscopy

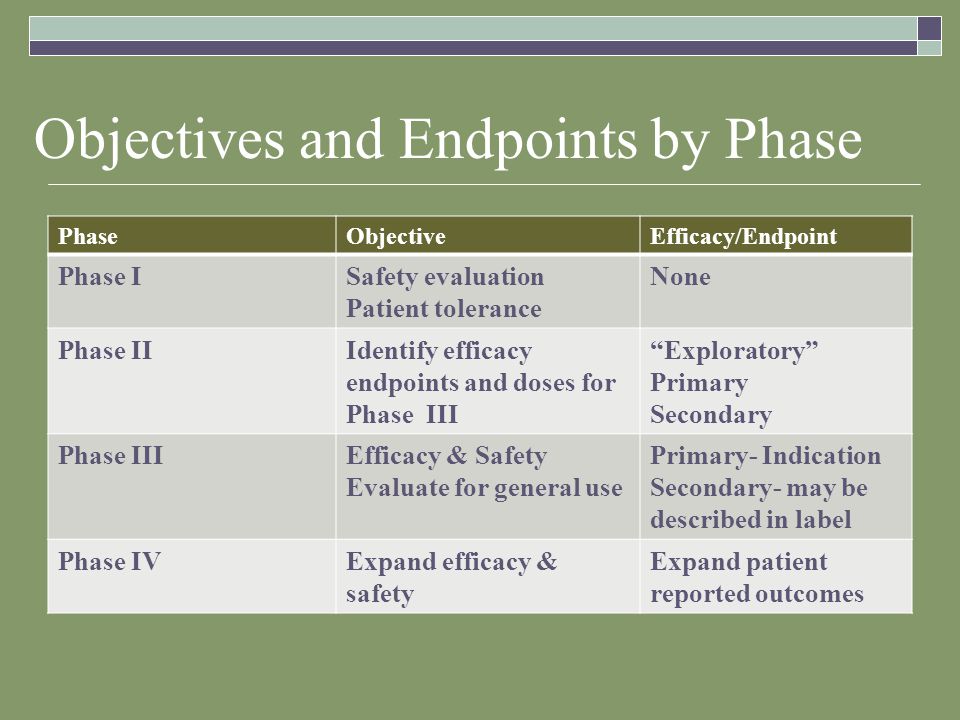
Development of Novel, Value-Based, Digital Endpoints for Clinical Trials: A Structured Approach Toward Fit-for-Purpose Validation | Pharmacological Reviews

Explanation of primary endpoints in acute myeloid leukemia clinical trials. | Download Scientific Diagram

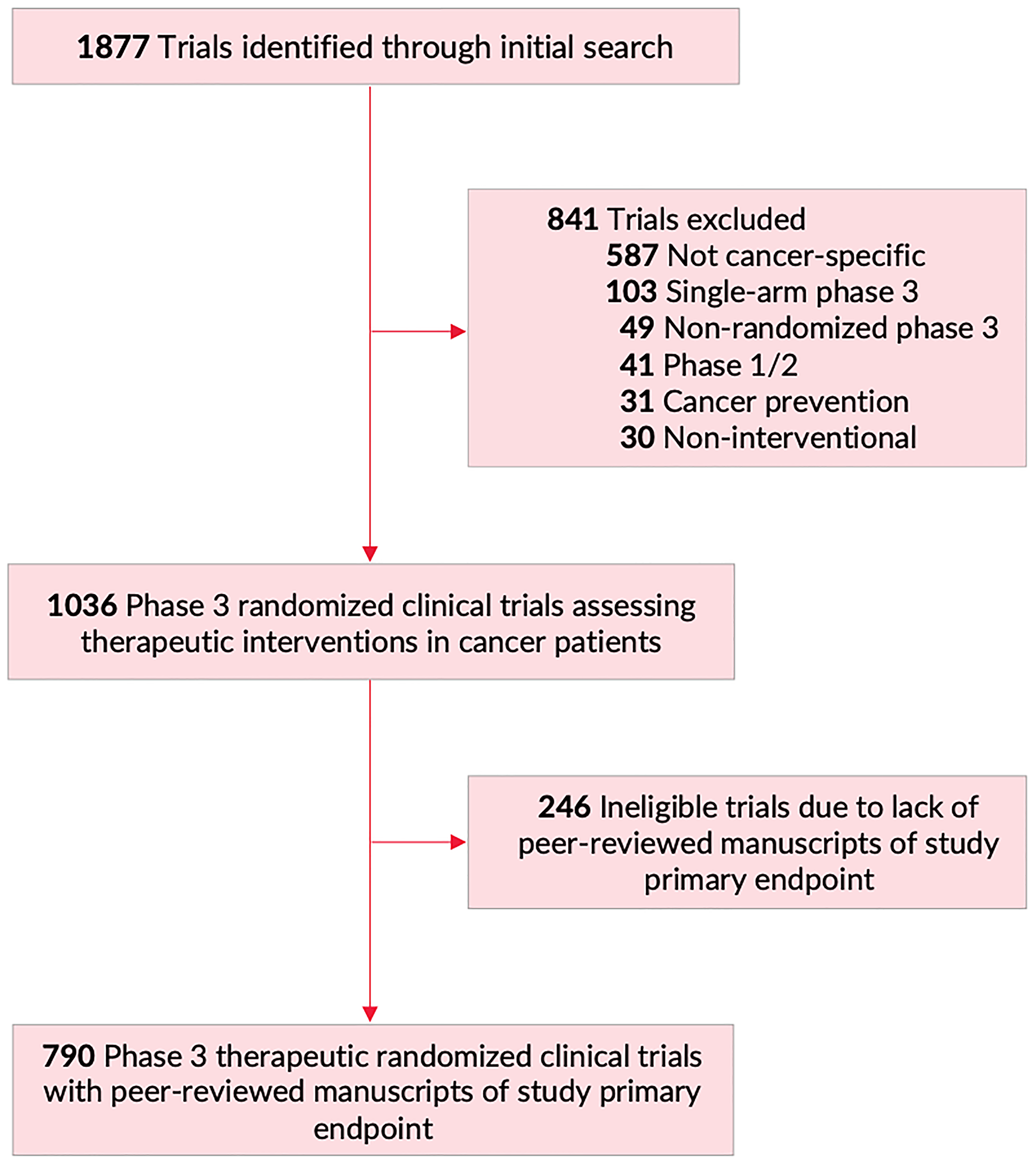
Selection of Endpoints in Clinical Trials: Trends in European Marketing Authorization Practice in Oncological Indications - Value in Health

Should Clinical Trial Interpretation Be Dominated by A P Value of 0.05 for the Primary Endpoint? | tctmd.com
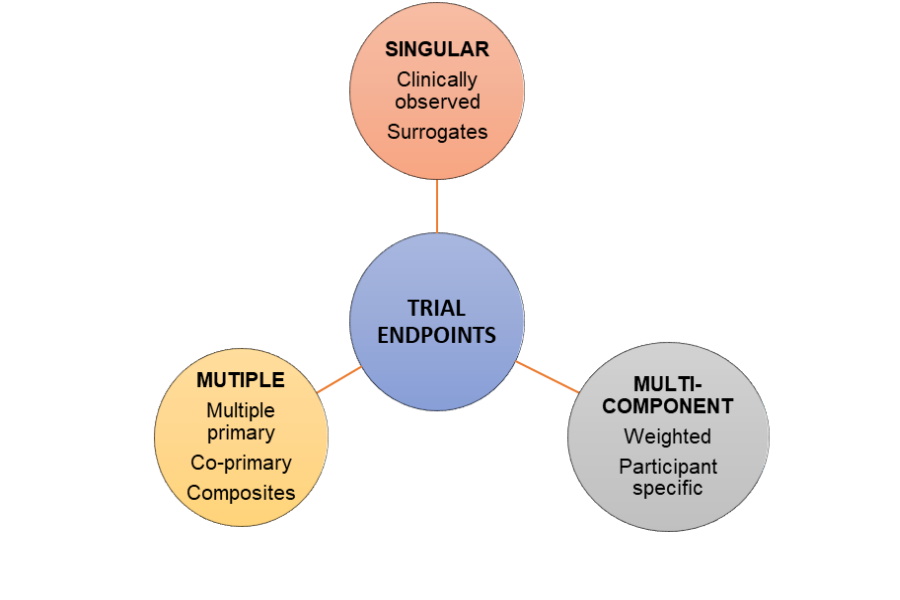
The Endpoint Selection: a Complex Process in the Clinical Trials Design Page CRA School | The International Clinical Research Academy Page | CRA School | The International Clinical Research Academy