Trends in patient‐reported outcome use in early phase dose‐finding oncology trials – an analysis of ClinicalTrials.gov - Lai‐Kwon - - Cancer Medicine - Wiley Online Library

Clinical Trial Patient-reported Outcomes Data: Going Beyond the Label in Oncology - Clinical Therapeutics

The impact of patient-reported outcome data from clinical trials: perspectives from international stakeholders | Journal of Patient-Reported Outcomes | Full Text

February 8, 2022: Patient-Centered Outcomes Core Highlights Lessons From the Demonstration Projects - Rethinking Clinical Trials

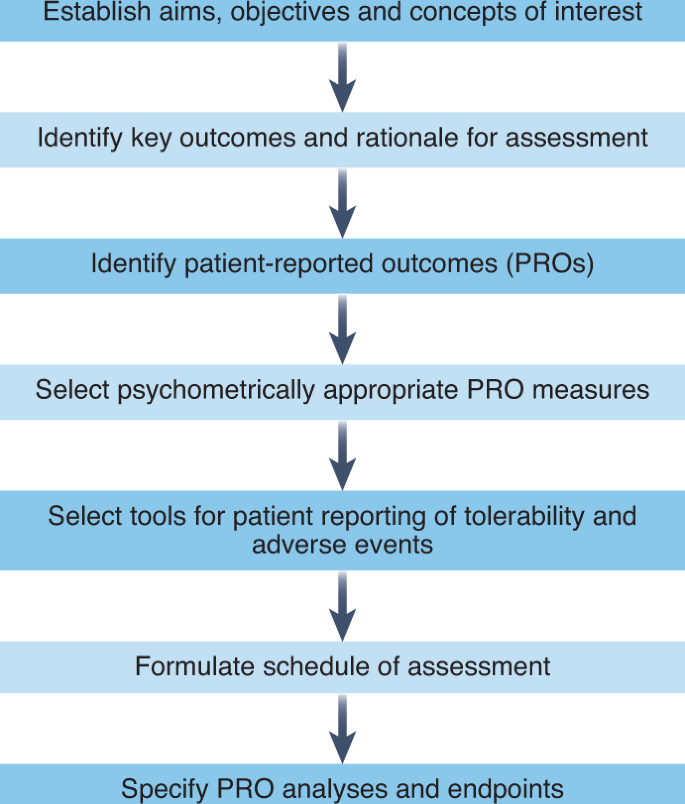
Systematic evaluation of patient-reported outcome (PRO) protocol content and reporting in UK cancer clinical trials: the EPiC study protocol | BMJ Open

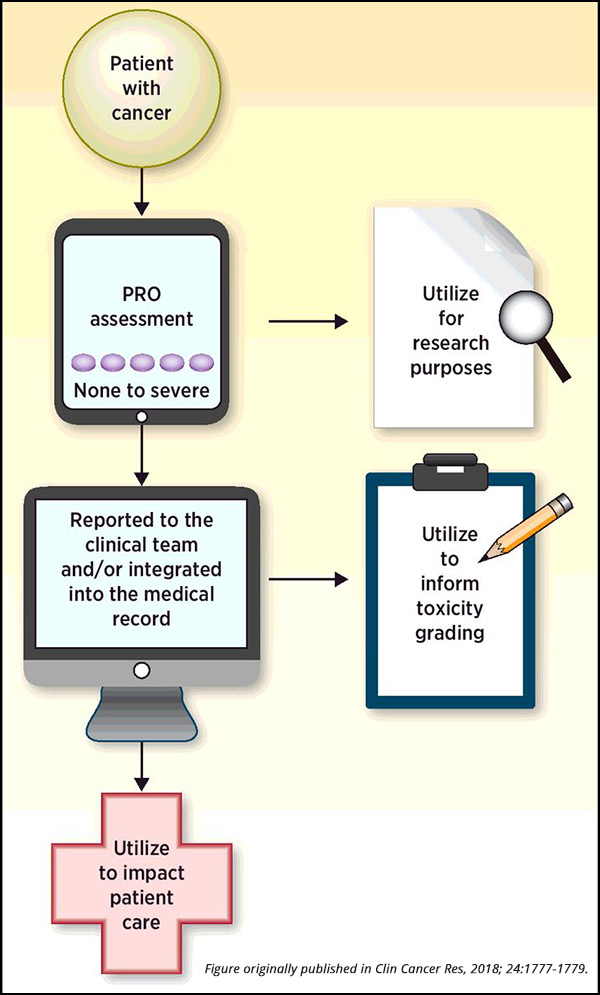
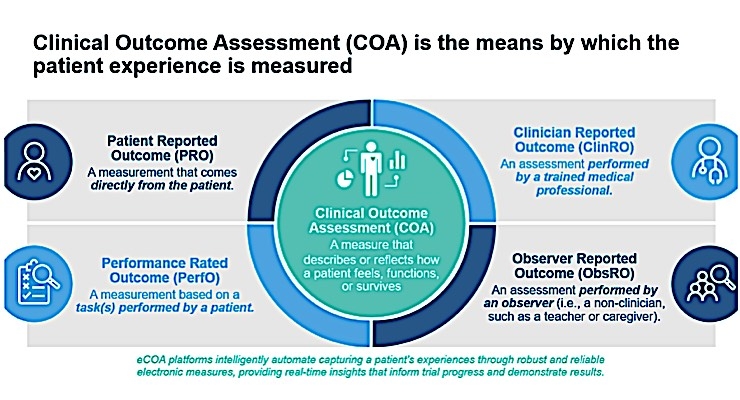
Methods for Implementing and Reporting Patient-reported Outcome (PRO) Measures of Symptomatic Adverse Events in Cancer Clinical Trials - Clinical Therapeutics
![PDF] Systematic evaluation of patient-reported outcome (PRO) protocol content and reporting in UK cancer clinical trials: the EPiC study protocol | Semantic Scholar PDF] Systematic evaluation of patient-reported outcome (PRO) protocol content and reporting in UK cancer clinical trials: the EPiC study protocol | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b14e4db6aa94553c20e782c6674e06150abbcc41/4-Figure1-1.png)
PDF] Systematic evaluation of patient-reported outcome (PRO) protocol content and reporting in UK cancer clinical trials: the EPiC study protocol | Semantic Scholar

References in Patient-Reported Outcomes as Primary End Points in Clinical Trials of Inflammatory Bowel Disease - Clinical Gastroenterology and Hepatology
PLOS ONE: Management of Patient-Reported Outcome (PRO) Alerts in Clinical Trials: A Cross Sectional Survey.

Table 2 from Patient Reported Outcomes (PROs) in Clinical Trials: Is 'In- Trial' Guidance Lacking? A Systematic Review | Semantic Scholar